ASH 2018: Is Venetoclax Effective Against High-Risk Multiple Myeloma?
Posted: Dec 08, 2018

Luciano J Costa, Associate Professor of Medicine Blood and Marrow Transplantation and Cell Therapy Program, University of Alabama Birmingham presented the results of a phase 2 clinical trial for Venetoclax in combination with Carfilzomib and Dexamethasone.
In 2016 the FDA approved Venclexta (venetoclax) for the treatment of patients with chronic lymphocytic leukemia (CLL) who have a chromosomal abnormality called 17p deletion. Later it was found to demonstrate activity in multiple myeloma. Venetoclax monotherapy showed activity among patients with relapsed or refractory multiple myeloma, particularly those with a translocation of t(11;14).
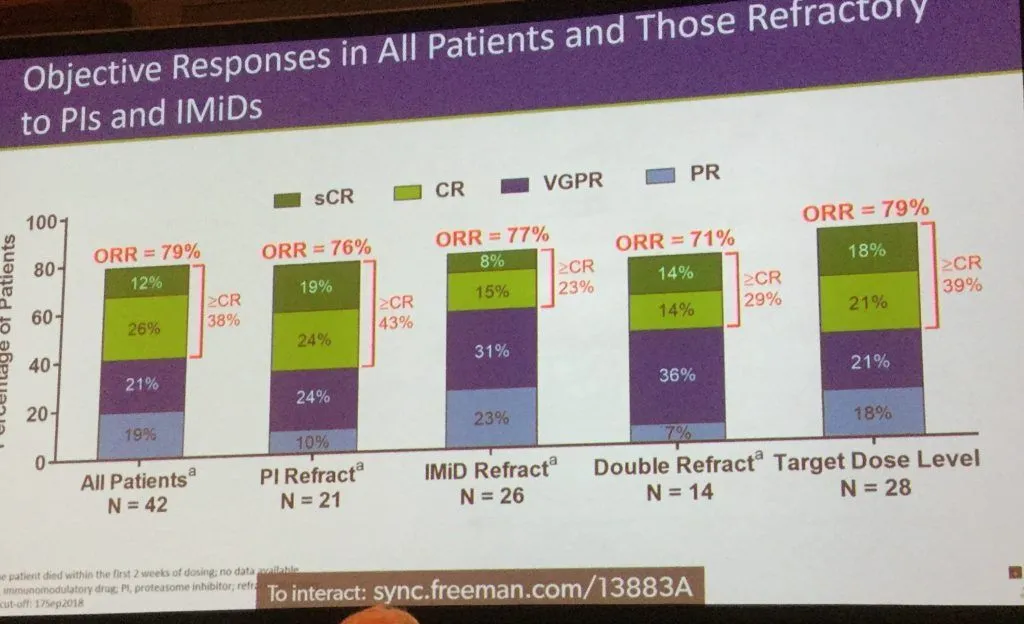
The overall response rate of 79% is remarkable, and is exceptional for patients who have relapsed after taking a proteasome inhibitor, immunomodulator or both.
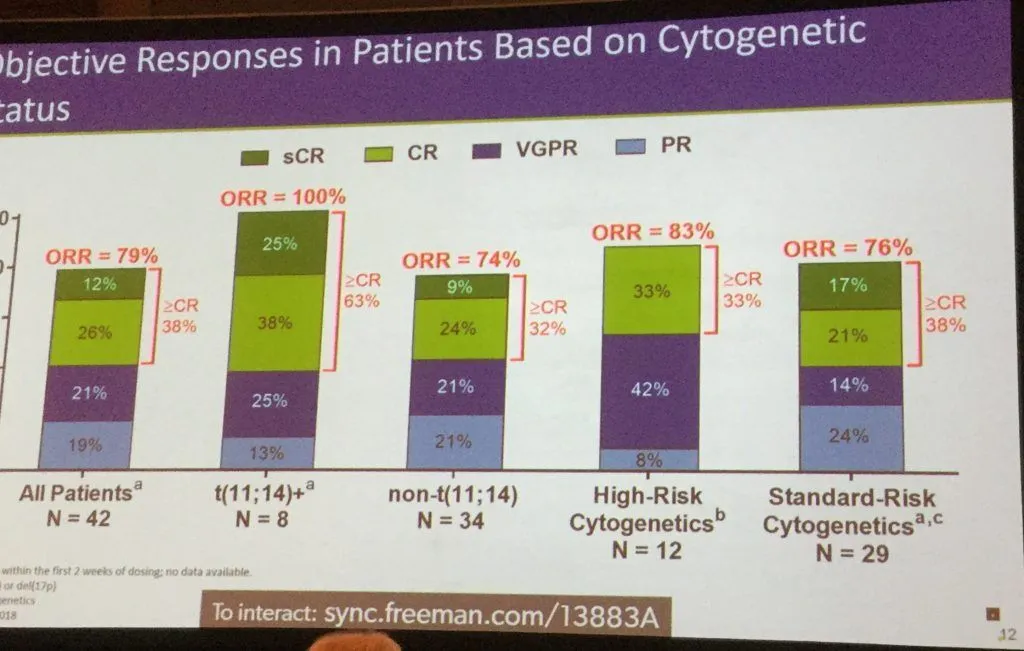
As in the single agent study, this combination has an excellent overall response rate for myeloma patients with a translocation of (11;14). A 100% response rate with 63% greater than complete response (CR). It shows outstanding results even in those with high risk cytogenetics (which includes the deletion of 17p). This a very important addition to the high risk treatment armamentarium.
Luciano J Costa, Associate Professor of Medicine Blood and Marrow Transplantation and Cell Therapy Program, University of Alabama Birmingham presented the results of a phase 2 clinical trial for Venetoclax in combination with Carfilzomib and Dexamethasone.
In 2016 the FDA approved Venclexta (venetoclax) for the treatment of patients with chronic lymphocytic leukemia (CLL) who have a chromosomal abnormality called 17p deletion. Later it was found to demonstrate activity in multiple myeloma. Venetoclax monotherapy showed activity among patients with relapsed or refractory multiple myeloma, particularly those with a translocation of t(11;14).
The overall response rate of 79% is remarkable, and is exceptional for patients who have relapsed after taking a proteasome inhibitor, immunomodulator or both.
As in the single agent study, this combination has an excellent overall response rate for myeloma patients with a translocation of (11;14). A 100% response rate with 63% greater than complete response (CR). It shows outstanding results even in those with high risk cytogenetics (which includes the deletion of 17p). This a very important addition to the high risk treatment armamentarium.

about the author
Gary Petersen
Gary is a myeloma survivor and patient advocate. His work centers around helping patients live longer by helping them to find facilities who are beating the average survival statistics. You can find Gary's site at www.myelomasurvival.com and follow him on Twitter at @grpetersen1
More on Treatment Advances
Thanks to our sponsors:


Get the Latest Multiple Myeloma Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.
100% of every dollar you give supports our life-changing mission.