Real-World Data on CAR T-Cell Therapy in Multiple Myeloma
CAR-T cell therapy has revolutionized hematologic cancer therapy and is on its way to becoming a broadly available myeloma treatment now that Abecma and Carvykti are FDA approved for relapsed myeloma patients.
In this therapy, T cells are extracted from the patient's blood and modified in the lab by adding a receptor (called a chimeric antigen receptor or CAR). Receptors can be thought of as a lock in a lock and key set. Once the locks are added this allows the T cells to detect individual cancer cell antigens (keys) more accurately. Then, these cells are infused back into the patient to attack malignant cells (Figure 1). 1
Figure 1. CAR T Cell procedure summary. 1
Since different cancers have different antigens (or keys), each CAR (or lock) is designed to target a unique cancer antigen. For example, Abecma is a CAR-T cell therapy recently approved by the FDA. T cells are genetically modified to recognize and attack myeloma by connecting your T cells to the B-cell maturation antigen (BCMA).2
Although this kind of therapy has shown remarkable efficacy in clinical trials,3 patients wonder about the impact it could have on their lives: How will my life be affected by a CAR-T cell treatment? What are real-world side effects that I should be worried about?
Nathan W. Sweeney, PhD and our team at HealthTree are researching these questions. They have analyzed clinical and patient-reported data from 17 patients who participated in CAR T-cell clinical trials (Table 1). 4
|
|
Table 1. Data analyzed in this study
Results
Our patients had a median of 10 lines of therapy before CAR T cell treatment. After CAR-T therapy, ten patients didn’t have new treatments to report at the time of the study. Five patients reported a new treatment, with a median time between treatments of 15.9 months. For two patients, there was no information available (Figure 2). This means that there was no need for new myeloma treatment after the CAR-T cell infusion in two-thirds of the patients. 4
Figure 2. The number of patients that reported new treatments after CAR-T cell therapy. 4
At the time of the study, the median time that our patients lived after CAR-T therapy (Overall Survival) was 24 months (21-30 months). Four of the 17 patients died with a median of 22.5 months post-CAR T-cell therapy, two of whom did not report a change in treatment. It is important to note that patients who have received CAR T therapy to date are highly relapsed patients with few options left.
Of the 17 patients, 13 patients (76%) reported myeloma reduction, three (18%) reported no myeloma reduction and one patient (6%) didn’t know his response (Figure 3). Almost four-fifths of the patients had myeloma reduction after CAR T-cell infusion.4
Figure 3. The number of patients that reported myeloma reduction after CAR-T cell therapy. 4
Patients reported 36 different side effects due to the CAR-T cell therapy. In figure 4, we have the most common side effects reported, all of which had a severity that varies from mild to moderate. The complete list and the corresponding severity can be found here. The side effects with the highest severity, reported by two patients each, can be found in table 2.4
Figure 4. The most common side effects reported by the patients. Those with a severity less than 3/10 are colored in green, and those with severity from 3 to 4/10 in yellow.4
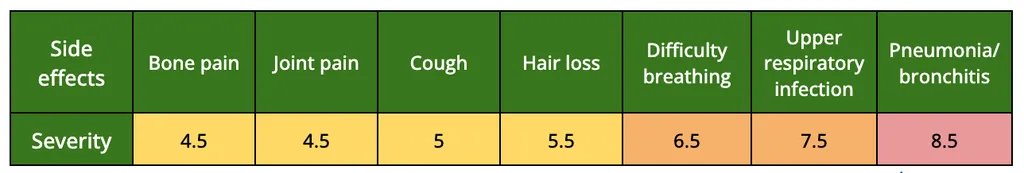
Table 2. The Side effects with the highest severity reported, using a 1 to 10 scale.4
Key Findings
- The data analyzed by Dr. Sweeney and our team at HealthTree suggests that CAR-T cell therapy is a viable option, with encouraging outcomes and manageable side effects for patients with difficult to control or relapsed myeloma
- Future directions in our research include analysis of genetics and treatment options post CAR T-cell therapy
- These results reinforce the importance of using patient-reported outcomes through an online platform, like HealthTree Cure Hub, to assess new therapeutics in the research community
References
- CAR T-Cells in Multiple Myeloma: State of the Art and Future Directions: https://www.frontiersin.org/articles/10.3389/fonc.2020.01243/full
- FDA Approves First Cell-Based Gene Therapy for Adult Patients with Multiple Myeloma: https://www.fda.gov/news-events/press-announcements/fda-approves-first-cell-based-gene-therapy-adult-patients-multiple-myeloma
- KarMMa-RW: comparison of idecabtagene vicleucel with real-world outcomes in relapsed and refractory multiple myeloma: https://www.nature.com/articles/s41408-021-00507-2
Patient-reported efficacy and toxicity in CAR T-cell therapy for multiple myeloma via Internet-based platforms. https://ascopubs.org/doi/10.1200/JCO.2021.39.15_suppl.e20024
CAR-T cell therapy has revolutionized hematologic cancer therapy and is on its way to becoming a broadly available myeloma treatment now that Abecma and Carvykti are FDA approved for relapsed myeloma patients.
In this therapy, T cells are extracted from the patient's blood and modified in the lab by adding a receptor (called a chimeric antigen receptor or CAR). Receptors can be thought of as a lock in a lock and key set. Once the locks are added this allows the T cells to detect individual cancer cell antigens (keys) more accurately. Then, these cells are infused back into the patient to attack malignant cells (Figure 1). 1
Figure 1. CAR T Cell procedure summary. 1
Since different cancers have different antigens (or keys), each CAR (or lock) is designed to target a unique cancer antigen. For example, Abecma is a CAR-T cell therapy recently approved by the FDA. T cells are genetically modified to recognize and attack myeloma by connecting your T cells to the B-cell maturation antigen (BCMA).2
Although this kind of therapy has shown remarkable efficacy in clinical trials,3 patients wonder about the impact it could have on their lives: How will my life be affected by a CAR-T cell treatment? What are real-world side effects that I should be worried about?
Nathan W. Sweeney, PhD and our team at HealthTree are researching these questions. They have analyzed clinical and patient-reported data from 17 patients who participated in CAR T-cell clinical trials (Table 1). 4
|
|
|
|||
|
|
|
|
|
|
Table 1. Data analyzed in this study
Results
Our patients had a median of 10 lines of therapy before CAR T cell treatment. After CAR-T therapy, ten patients didn’t have new treatments to report at the time of the study. Five patients reported a new treatment, with a median time between treatments of 15.9 months. For two patients, there was no information available (Figure 2). This means that there was no need for new myeloma treatment after the CAR-T cell infusion in two-thirds of the patients. 4
Figure 2. The number of patients that reported new treatments after CAR-T cell therapy. 4
At the time of the study, the median time that our patients lived after CAR-T therapy (Overall Survival) was 24 months (21-30 months). Four of the 17 patients died with a median of 22.5 months post-CAR T-cell therapy, two of whom did not report a change in treatment. It is important to note that patients who have received CAR T therapy to date are highly relapsed patients with few options left.
Of the 17 patients, 13 patients (76%) reported myeloma reduction, three (18%) reported no myeloma reduction and one patient (6%) didn’t know his response (Figure 3). Almost four-fifths of the patients had myeloma reduction after CAR T-cell infusion.4
Figure 3. The number of patients that reported myeloma reduction after CAR-T cell therapy. 4
Patients reported 36 different side effects due to the CAR-T cell therapy. In figure 4, we have the most common side effects reported, all of which had a severity that varies from mild to moderate. The complete list and the corresponding severity can be found here. The side effects with the highest severity, reported by two patients each, can be found in table 2.4
Figure 4. The most common side effects reported by the patients. Those with a severity less than 3/10 are colored in green, and those with severity from 3 to 4/10 in yellow.4

Table 2. The Side effects with the highest severity reported, using a 1 to 10 scale.4
Key Findings
- The data analyzed by Dr. Sweeney and our team at HealthTree suggests that CAR-T cell therapy is a viable option, with encouraging outcomes and manageable side effects for patients with difficult to control or relapsed myeloma
- Future directions in our research include analysis of genetics and treatment options post CAR T-cell therapy
- These results reinforce the importance of using patient-reported outcomes through an online platform, like HealthTree Cure Hub, to assess new therapeutics in the research community
References
- CAR T-Cells in Multiple Myeloma: State of the Art and Future Directions: https://www.frontiersin.org/articles/10.3389/fonc.2020.01243/full
- FDA Approves First Cell-Based Gene Therapy for Adult Patients with Multiple Myeloma: https://www.fda.gov/news-events/press-announcements/fda-approves-first-cell-based-gene-therapy-adult-patients-multiple-myeloma
- KarMMa-RW: comparison of idecabtagene vicleucel with real-world outcomes in relapsed and refractory multiple myeloma: https://www.nature.com/articles/s41408-021-00507-2
Patient-reported efficacy and toxicity in CAR T-cell therapy for multiple myeloma via Internet-based platforms. https://ascopubs.org/doi/10.1200/JCO.2021.39.15_suppl.e20024

about the author
Arturo Hurtado
Arturo Hurtado is an International Medical Graduate who Joined HealthTree in 2020 as part of The Patient Experience team. He helps patients understand their disease panorama and navigate their myeloma through the tools and resources that HealthTree provides. He is an enthusiastic photographer, tech nerd, and aspiring food explorer who loves to travel and find new exciting experiences.
More on HealthTree Research


Get the Latest Multiple Myeloma Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.












