Blood Based MRD Testing Advances in Multiple Myeloma During COVID-19
As myeloma patients reduce their visits and regular testing to protect themselves from contracting COVID-19, they are likely to also reduce the number of bone marrow biopsies they receive. This might mean that less minimal residual disease (MRD) testing is being performed.
MRD testing is used to determine the level of disease following myeloma therapy. It is a more sensitve way of testing very low levels of myeloma that go beyond today's blood and bone marrow biopsy tests. MRD tests can use either a flow cytometry or Next Generation Sequencing methodologies. (Learn more about MRD here). MRD testing today is performed on a bone marrow biopsy sample.
So can a blood test perform adequate MRD testing? Adaptive Biotechnologies is working on a new test to do just that.
Adaptive’s laboratory in Seattle offers blood-based MRD testing as a CLIA-regulated laboratory-developed test service (Clinical Laboratory Improvement Amendments). This test is can be run on patients with ALL, CLL and multiple myeloma. The clonoSEQ MRD test is currently covered by Medicare and many private insurers.
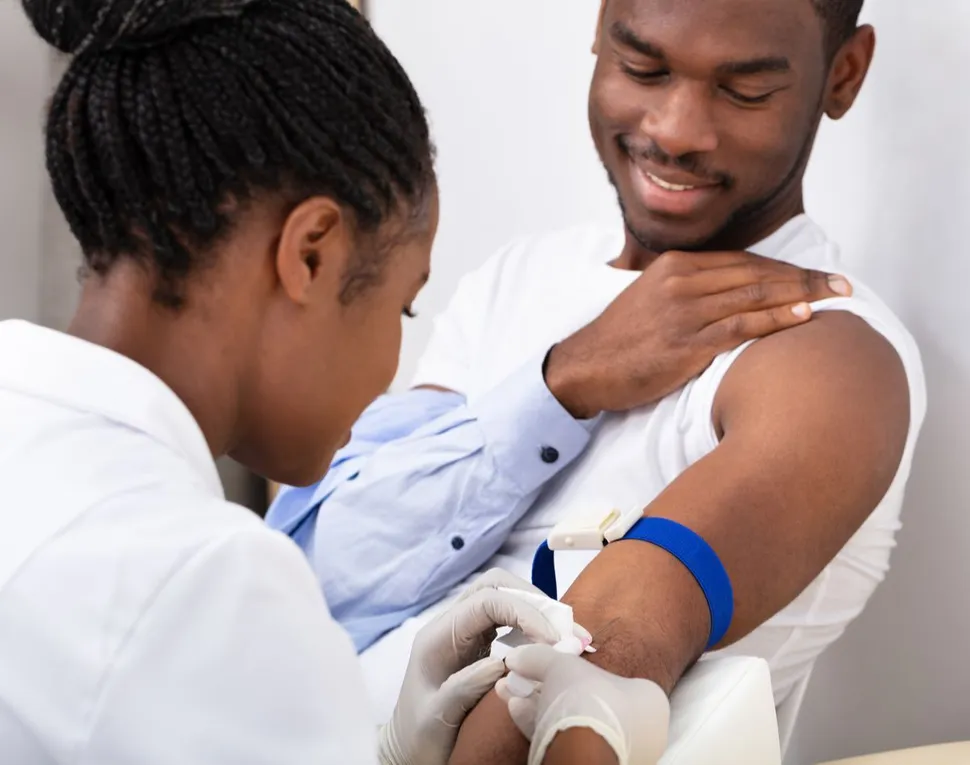
You can ask your doctor if he/she can draw an extra 2mL of blood at your next appointment and ship the sample overnight (unrefrigerated) to the Seattle lab. This is still an early test under review by the FDA. The bone marrow-based clonoSEQ test is FDA approved while the blood-based clonoSEQ test is not yet approved.
Future studies will compare the blood vs. bone marrow results to determine if the tests are similar in sensitivity.
As myeloma patients reduce their visits and regular testing to protect themselves from contracting COVID-19, they are likely to also reduce the number of bone marrow biopsies they receive. This might mean that less minimal residual disease (MRD) testing is being performed.
MRD testing is used to determine the level of disease following myeloma therapy. It is a more sensitve way of testing very low levels of myeloma that go beyond today's blood and bone marrow biopsy tests. MRD tests can use either a flow cytometry or Next Generation Sequencing methodologies. (Learn more about MRD here). MRD testing today is performed on a bone marrow biopsy sample.
So can a blood test perform adequate MRD testing? Adaptive Biotechnologies is working on a new test to do just that.
Adaptive’s laboratory in Seattle offers blood-based MRD testing as a CLIA-regulated laboratory-developed test service (Clinical Laboratory Improvement Amendments). This test is can be run on patients with ALL, CLL and multiple myeloma. The clonoSEQ MRD test is currently covered by Medicare and many private insurers.
You can ask your doctor if he/she can draw an extra 2mL of blood at your next appointment and ship the sample overnight (unrefrigerated) to the Seattle lab. This is still an early test under review by the FDA. The bone marrow-based clonoSEQ test is FDA approved while the blood-based clonoSEQ test is not yet approved.
Future studies will compare the blood vs. bone marrow results to determine if the tests are similar in sensitivity.
about the author
Jennifer Ahlstrom
Myeloma survivor, patient advocate, wife, mom of 6. Believer that patients can contribute to cures by joining HealthTree Cure Hub and joining clinical research. Founder and CEO of HealthTree Foundation.
More on Treatment Advances
Trending Articles




Get the Latest Multiple Myeloma Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.