The Dawning Era of Bispecific Antibodies in Myeloma

Bispecific antibodies, also known as T-cell engagers or T-cell redirective therapy, are showing great promise in relapsed/refractory myeloma. Bispecific antibodies are antibodies that have two arms. One of the arms is drawn to a target antigen, such as BCMA or GPRC5D, which is located on the myeloma cell. The other arm is drawn to CD3 found on the T-cell.
The idea is if the bispecific antibody is able to “redirect” the T-cell to the myeloma cell then the T-cell will be able to kill the myeloma. What an ingenious idea! Check this video out to learn how bispecific antibodies work.
Let’s take a look at some of the latest data that has been shared recently at the American Society of Clinical Oncology’s (ASCO) annual meeting and the annual meeting of the European Hematological Association (EHA) to see why I am very hopefully for the future.
Teclistamab
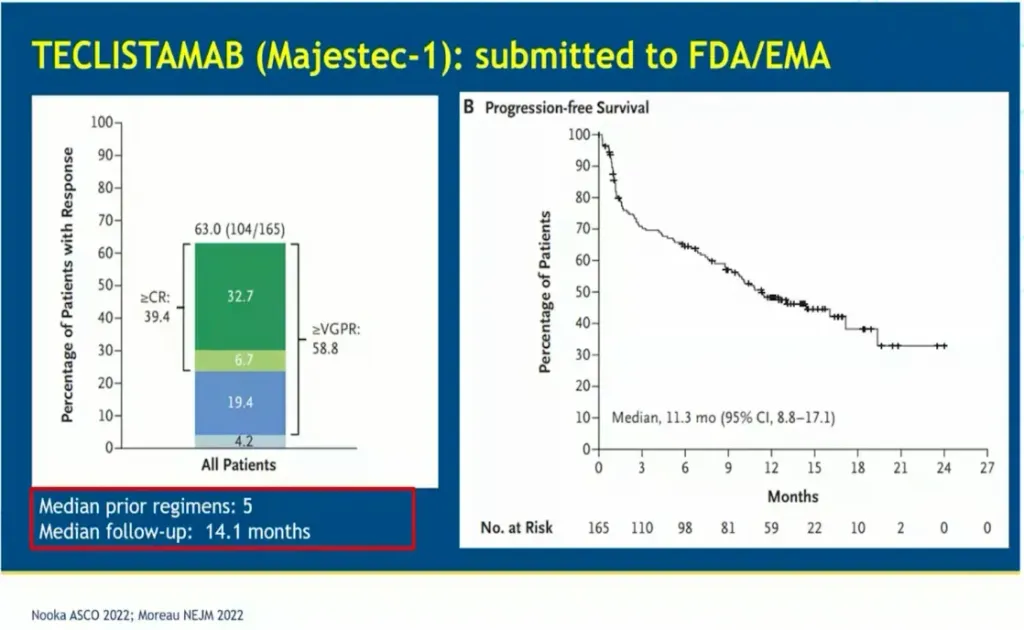
Results from the MajesTEC (TEClistamab) trial showed an overall response rate of 63% which is very good for this heavily pretreated population. A significant number (27%) of the trial participants became MRD negative. The major side effects of teclistamab are cytokine release syndrome (CRS) and neurotoxicity (ICANS). Many (70%) of the trial participants experienced grade 1 or 2 CRS and 3% experienced grade 1 or 2 ICANS. The median progression free survival (PFS) was reported to be 11.3 months.
Elranatamab
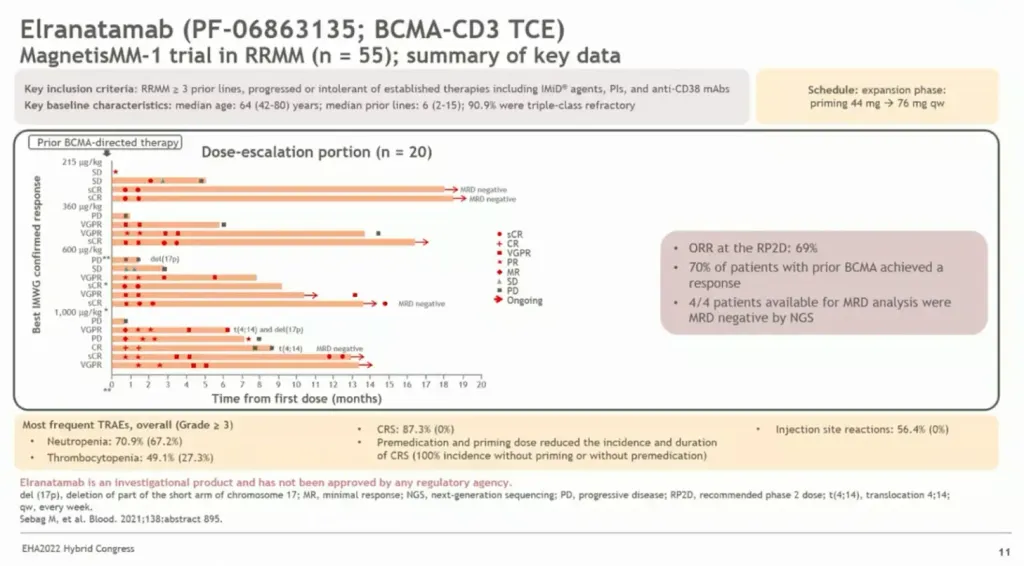
In the MagnetisMM trial, elranatamab, a subcutaneous BCMA-targeted T-cell redirecting immunotherapy (AKA bispecific antibody), for patients with relapsed or refractory multiple myeloma also showed nice results. Elranatamab shows an overall response rate of 54%. Sixty-seven (67%) percent of the trial participants experienced grade 1 or 2 CRS. The percentage of the participants that experienced grade 1 or 2 ICANs was in the single digits. The median progression free survival (PFS) has not been reached at a median follow-up of 8.1 months. In the BCMA exposed/refractory myeloma patients, elranatamab shows a response rate around 60%, with follow-up of about 4 months.
Talquetamab
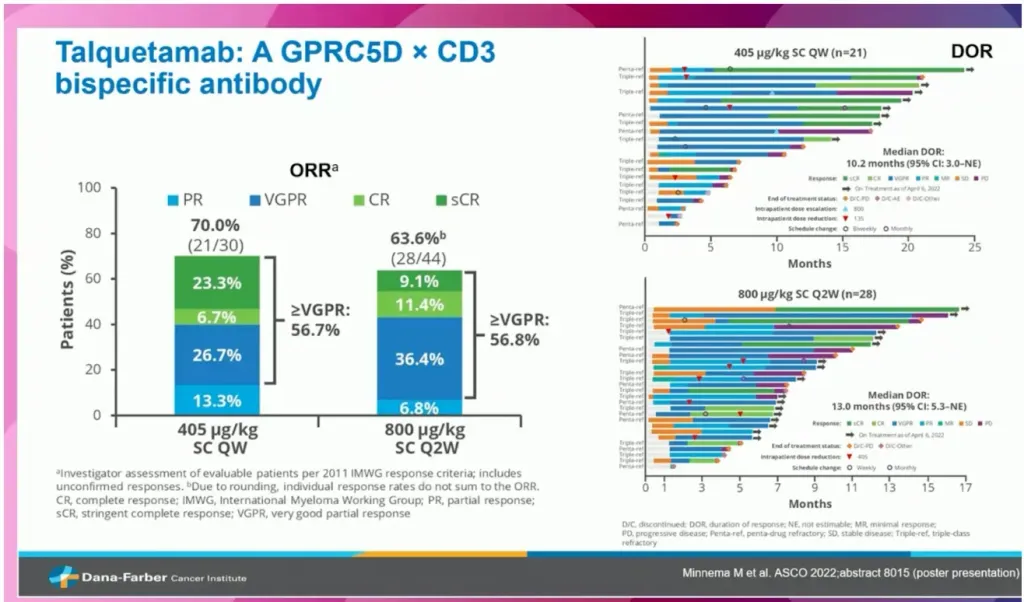
In the MonumenTAL study, TALquetamab, a subcutaneous bispecific antibody that targets GPRC5D, showed an overall response rate of 70% at the 405mcg/kg dose and 64% at the 800 mcg/kg dose. Twenty-seven percent (27%) of the trial participants became MRD negative. Besides CRS, skin and nail related adverse events were seen in 83% of patients at grade 1 and 75% of patients at grade 2. Also, 63% of patients at grade 1 and 57% of patients at grade experienced “dysgeusia” which means foods tasted sour, metallic or bitter. The median follow-up is 11.7 months.
REGN5458
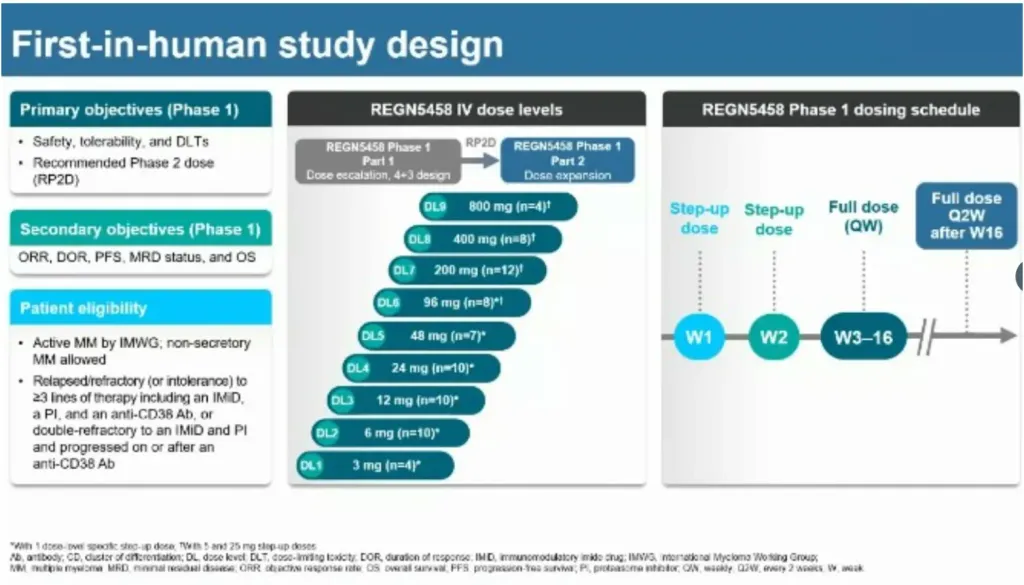
REGN5458, an IV bispecific antibody directed at BCMA, showed an overall response rate of 50.7% and a 75% response rate in the 200-800 mg dose group. It has the lowest CRS amongst the bispecific antibodies so far ~38.4% (grade 1/2) with a median follow-up of 3 months.
Talquetamab with Darzalex
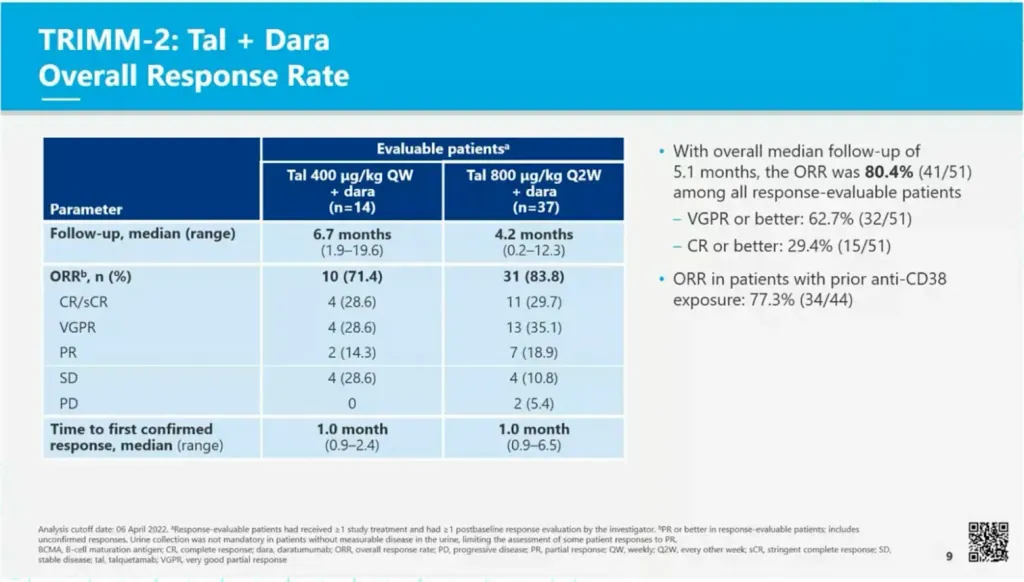
T-cell redirecting therapies (bispecific antibodies) are now being used in combination with other anti-myeloma agents. In the TRiMM-2 study, (my inside sources tell me that TRiMM stands for T-cell Redirection in Multiple Myeloma), Talquetamab is being combined with daratumumab (Darzalex). With an overall median follow up of 5.1 months, the overall response rate was 80.4% in the relapsed/refractory patient population, the majority who were refractory to anti-CD38 therapies (daratumumab or isatuximab). There was no grade 3 or 4 CRS observed. Two patients had a grade 1 ICANS that resolved within a day.
The era of T-cell redirecting therapies has dawned.
To get up to speed with the latest happenings in myeloma research, view our Immunotherapy Units and guest lectures in HealthTree University.
Bispecific antibodies, also known as T-cell engagers or T-cell redirective therapy, are showing great promise in relapsed/refractory myeloma. Bispecific antibodies are antibodies that have two arms. One of the arms is drawn to a target antigen, such as BCMA or GPRC5D, which is located on the myeloma cell. The other arm is drawn to CD3 found on the T-cell.
The idea is if the bispecific antibody is able to “redirect” the T-cell to the myeloma cell then the T-cell will be able to kill the myeloma. What an ingenious idea! Check this video out to learn how bispecific antibodies work.
Let’s take a look at some of the latest data that has been shared recently at the American Society of Clinical Oncology’s (ASCO) annual meeting and the annual meeting of the European Hematological Association (EHA) to see why I am very hopefully for the future.
Teclistamab
Results from the MajesTEC (TEClistamab) trial showed an overall response rate of 63% which is very good for this heavily pretreated population. A significant number (27%) of the trial participants became MRD negative. The major side effects of teclistamab are cytokine release syndrome (CRS) and neurotoxicity (ICANS). Many (70%) of the trial participants experienced grade 1 or 2 CRS and 3% experienced grade 1 or 2 ICANS. The median progression free survival (PFS) was reported to be 11.3 months.

Elranatamab
In the MagnetisMM trial, elranatamab, a subcutaneous BCMA-targeted T-cell redirecting immunotherapy (AKA bispecific antibody), for patients with relapsed or refractory multiple myeloma also showed nice results. Elranatamab shows an overall response rate of 54%. Sixty-seven (67%) percent of the trial participants experienced grade 1 or 2 CRS. The percentage of the participants that experienced grade 1 or 2 ICANs was in the single digits. The median progression free survival (PFS) has not been reached at a median follow-up of 8.1 months. In the BCMA exposed/refractory myeloma patients, elranatamab shows a response rate around 60%, with follow-up of about 4 months.

Talquetamab
In the MonumenTAL study, TALquetamab, a subcutaneous bispecific antibody that targets GPRC5D, showed an overall response rate of 70% at the 405mcg/kg dose and 64% at the 800 mcg/kg dose. Twenty-seven percent (27%) of the trial participants became MRD negative. Besides CRS, skin and nail related adverse events were seen in 83% of patients at grade 1 and 75% of patients at grade 2. Also, 63% of patients at grade 1 and 57% of patients at grade experienced “dysgeusia” which means foods tasted sour, metallic or bitter. The median follow-up is 11.7 months.

REGN5458
REGN5458, an IV bispecific antibody directed at BCMA, showed an overall response rate of 50.7% and a 75% response rate in the 200-800 mg dose group. It has the lowest CRS amongst the bispecific antibodies so far ~38.4% (grade 1/2) with a median follow-up of 3 months.

Talquetamab with Darzalex
T-cell redirecting therapies (bispecific antibodies) are now being used in combination with other anti-myeloma agents. In the TRiMM-2 study, (my inside sources tell me that TRiMM stands for T-cell Redirection in Multiple Myeloma), Talquetamab is being combined with daratumumab (Darzalex). With an overall median follow up of 5.1 months, the overall response rate was 80.4% in the relapsed/refractory patient population, the majority who were refractory to anti-CD38 therapies (daratumumab or isatuximab). There was no grade 3 or 4 CRS observed. Two patients had a grade 1 ICANS that resolved within a day.

The era of T-cell redirecting therapies has dawned.
To get up to speed with the latest happenings in myeloma research, view our Immunotherapy Units and guest lectures in HealthTree University.

about the author
Cynthia Chmielewski
Cynthia (Cindy) Chmielewski is a professional educator and myeloma advocate. As a former teacher, she now teaches myeloma patients how to advocate for themselves as the Director of HealthTree University. You can follow her on Twitter @myelomateacher
More on HealthTree Research
Trending Articles

Get the Latest Multiple Myeloma Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.












