Clinical Trial: Phase 1 BCMA-CD3 Bispecific Antibody for Relapsed/Refractory Myeloma Patients
A new Phase 1 study is open to test different doses of a new bispecific antibody treatment for patients with relapsed/refractory multiple myeloma.
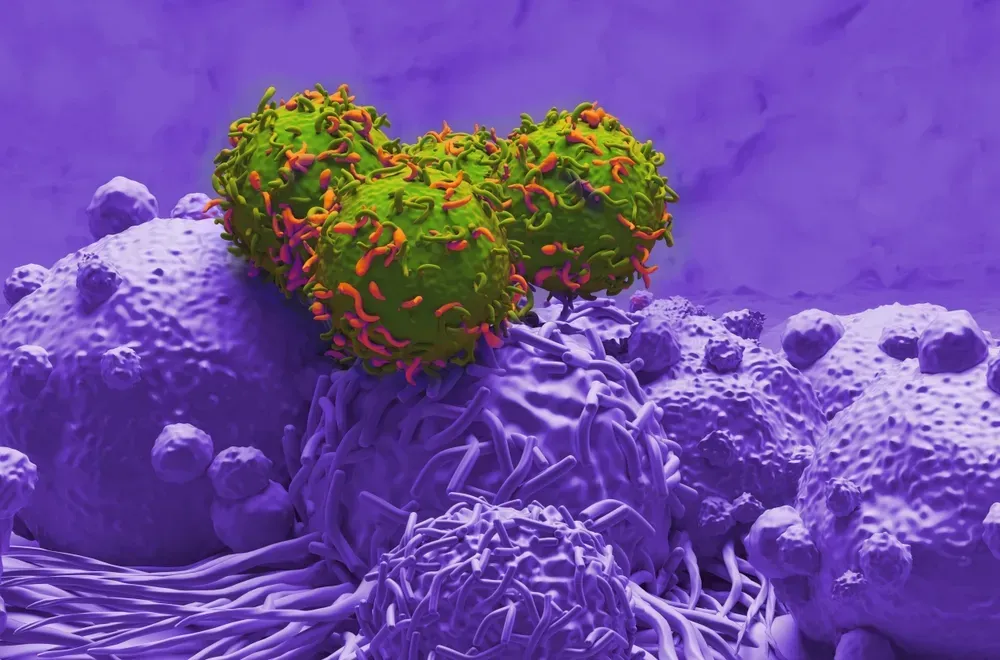
The new drug (called PF-06863135) is a bispecific antibody, which is a new treatment designed to have two different targets. In this case, PF-06863135 targets CD3, which is found on T Cells, and BCMA, which may be expressed by myeloma cells. PF-06863135 draws them together, which allows the T cell to better target and potentially kill the myeloma cell.
This trial is seeking an estimated 80 patients, and will be testing dose escalation, followed by dose expansion to assess overall response and duration of response. This trial is currently open and accepting patients at multiple locations in the US.
Patients must have relapsed/refractory multiple myeloma, and adequate bone marrow, kidney and liver function.
Click the link below to learn more about this study and see if you are eligible. You can also call SparkCures with any questions at (888) 828-2206.
To learn more, find this study on SparkCures here:
Phase 1 BCMA-CD3 Bispecific Antibody Trial
You can learn more about bispecific antibodies in a prior Myeloma Crowd article here.
A new Phase 1 study is open to test different doses of a new bispecific antibody treatment for patients with relapsed/refractory multiple myeloma.
The new drug (called PF-06863135) is a bispecific antibody, which is a new treatment designed to have two different targets. In this case, PF-06863135 targets CD3, which is found on T Cells, and BCMA, which may be expressed by myeloma cells. PF-06863135 draws them together, which allows the T cell to better target and potentially kill the myeloma cell.
This trial is seeking an estimated 80 patients, and will be testing dose escalation, followed by dose expansion to assess overall response and duration of response. This trial is currently open and accepting patients at multiple locations in the US.
Patients must have relapsed/refractory multiple myeloma, and adequate bone marrow, kidney and liver function.
Click the link below to learn more about this study and see if you are eligible. You can also call SparkCures with any questions at (888) 828-2206.
To learn more, find this study on SparkCures here:
Phase 1 BCMA-CD3 Bispecific Antibody Trial
You can learn more about bispecific antibodies in a prior Myeloma Crowd article here.
about the author
Jennifer Ahlstrom
Myeloma survivor, patient advocate, wife, mom of 6. Believer that patients can contribute to cures by joining HealthTree Cure Hub and joining clinical research. Founder and CEO of HealthTree Foundation.
More on Navigating Your Health
Trending Articles




Get the Latest Multiple Myeloma Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.
3x Faster.