Myeloma CAR-T Achieves 100% Response Rate in High-Risk Patients

A Phase I Single-Arm Study of a new CAR-T product for myeloma, known as GC012F resulted in a 100% response rate for each of the 22 patients within the clinical trial. Although 22 is not a large number, the potential of this therapy is exciting, especially because each of the 22 patients had high-risk multiple myeloma and were newly diagnosed.
As you may know, thanks to immunotherapies and other new drugs on the myeloma market, the average longevity of a myeloma patient's life has exponentially increased. Where most patients were living 3-5 years just a decade ago, now standard-risk myeloma patients are often seeing results that last for a decade or longer. Treatment is more effective and dosages are less toxic, allowing patients to enjoy a high-quality life that doesn't involve myeloma in every waking moment.
However, that success has not been the same for high-risk myeloma patients. While certainly their lives are improved due to these immunotherapies and other advancements in the myeloma field, they have less sustainable responses and can often progress quicker than their standard-risk counterparts, leaving them to burn through these successful therapies quicker than the average myeloma patient.
This causes a lot of stress for high-risk patients, caregivers, and physicians alike who have great tools at their disposal but don't want to feel like time is running out.
This dual-targeting CAR-T therapy has the potential to be another option for high-risk myeloma treatment, and one that is seeing promising early results.
About the Dual-Targeting CAR T-cell Product
This new CAR-T product is exciting for many reasons.
- It's dual targeting. Current FDA-approved CAR-T products like Abecma or CARVYKTI only have one target, BCMA. This new CAR-T therapy, GC012F, targets both BCMA and CD-19 on the myeloma cell, making it more likely to identify and eradicate multiple myeloma cells (vs other cells) in the immune system.
- It's a FasTCAR-T product. While the myeloma CAR-Ts that we currently have on the market have to be sent to a lab to be re-engineered in a process that can take up to two weeks, this product is part of a new generation of CARs in which they can re-generate quicker, sometimes within one to two days.
Exciting Trial Results for Myeloma-Targeting GC012F
This was a ground-breaking trial with a lot of potential, which you can see below.
- All 22 high-risk myeloma patients in this trial achieved a response, with 95.5% of patients achieving sCR (stringent complete response).
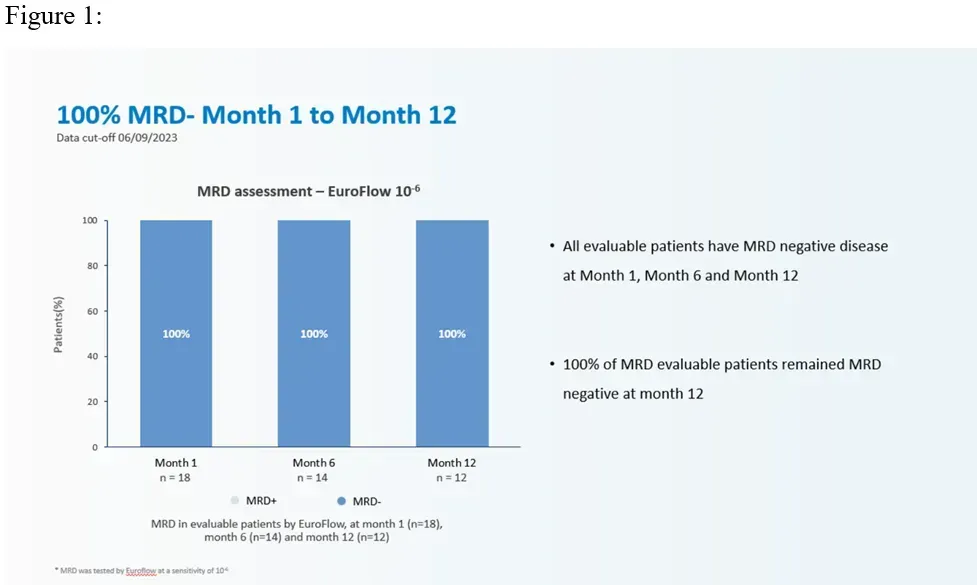
- All treated pts (100%) across all dose levels achieved minimal residual disease (MRD) negativity assessed by Euroflow (sensitivity of 10-6).
- Only 6 pts (27%) experience low-grade cytokine release syndrome (CRS), including 23% grade 1 (n=5) and 4% grade 2 (n=1). No treatment-related grade ≥3 CRS (which would require an ICU stay), nor ICANS (neurotoxicity) of any grade, nor any deaths occurred in the study. This indicates that this CAR-T might have a less risky side effect profile than the CAR-T products that are currently on the market. A larger data set of myeloma patients on a similar trial would confirm this.
Sustainable Results for High-Risk Multiple Myeloma Patients
Perhaps the most important outcome of this trial was the durability of response in the high-risk multiple myeloma patients. As mentioned above, it happens all too often that high-risk myeloma patients achieve significantly less durable outcomes than their standard-risk counterparts on novel and immunotherapies. However, this trial showed that not only did ALL of the patients respond to this CAR-T product, but a high number of these patients were able to sustain their responses for over a year.
You can see this demonstrated in the image below.
Conclusion
This dual-targeting CAR T-cell therapy contains a lot of potential and promise. The company that produces the CAR-T, Gracell Biotechnologies, was recently bought by AztraZeneca, a pharma company.
“GC012F will accelerate our cell therapy strategy in haematology, with the opportunity to bring a potential best-in-class treatment to patients living with blood cancers using a differentiated manufacturing process,” Susan Galbraith, MD, PhD, executive vice president of AstraZeneca’s oncology R&D."
The development of this product, and certainly more clinical trials with the product, will continue to move forward.
A Phase I Single-Arm Study of a new CAR-T product for myeloma, known as GC012F resulted in a 100% response rate for each of the 22 patients within the clinical trial. Although 22 is not a large number, the potential of this therapy is exciting, especially because each of the 22 patients had high-risk multiple myeloma and were newly diagnosed.
As you may know, thanks to immunotherapies and other new drugs on the myeloma market, the average longevity of a myeloma patient's life has exponentially increased. Where most patients were living 3-5 years just a decade ago, now standard-risk myeloma patients are often seeing results that last for a decade or longer. Treatment is more effective and dosages are less toxic, allowing patients to enjoy a high-quality life that doesn't involve myeloma in every waking moment.
However, that success has not been the same for high-risk myeloma patients. While certainly their lives are improved due to these immunotherapies and other advancements in the myeloma field, they have less sustainable responses and can often progress quicker than their standard-risk counterparts, leaving them to burn through these successful therapies quicker than the average myeloma patient.
This causes a lot of stress for high-risk patients, caregivers, and physicians alike who have great tools at their disposal but don't want to feel like time is running out.
This dual-targeting CAR-T therapy has the potential to be another option for high-risk myeloma treatment, and one that is seeing promising early results.
About the Dual-Targeting CAR T-cell Product
This new CAR-T product is exciting for many reasons.
- It's dual targeting. Current FDA-approved CAR-T products like Abecma or CARVYKTI only have one target, BCMA. This new CAR-T therapy, GC012F, targets both BCMA and CD-19 on the myeloma cell, making it more likely to identify and eradicate multiple myeloma cells (vs other cells) in the immune system.
- It's a FasTCAR-T product. While the myeloma CAR-Ts that we currently have on the market have to be sent to a lab to be re-engineered in a process that can take up to two weeks, this product is part of a new generation of CARs in which they can re-generate quicker, sometimes within one to two days.
Exciting Trial Results for Myeloma-Targeting GC012F
This was a ground-breaking trial with a lot of potential, which you can see below.
- All 22 high-risk myeloma patients in this trial achieved a response, with 95.5% of patients achieving sCR (stringent complete response).
- All treated pts (100%) across all dose levels achieved minimal residual disease (MRD) negativity assessed by Euroflow (sensitivity of 10-6).
- Only 6 pts (27%) experience low-grade cytokine release syndrome (CRS), including 23% grade 1 (n=5) and 4% grade 2 (n=1). No treatment-related grade ≥3 CRS (which would require an ICU stay), nor ICANS (neurotoxicity) of any grade, nor any deaths occurred in the study. This indicates that this CAR-T might have a less risky side effect profile than the CAR-T products that are currently on the market. A larger data set of myeloma patients on a similar trial would confirm this.
Sustainable Results for High-Risk Multiple Myeloma Patients
Perhaps the most important outcome of this trial was the durability of response in the high-risk multiple myeloma patients. As mentioned above, it happens all too often that high-risk myeloma patients achieve significantly less durable outcomes than their standard-risk counterparts on novel and immunotherapies. However, this trial showed that not only did ALL of the patients respond to this CAR-T product, but a high number of these patients were able to sustain their responses for over a year.
You can see this demonstrated in the image below.
Conclusion
This dual-targeting CAR T-cell therapy contains a lot of potential and promise. The company that produces the CAR-T, Gracell Biotechnologies, was recently bought by AztraZeneca, a pharma company.
“GC012F will accelerate our cell therapy strategy in haematology, with the opportunity to bring a potential best-in-class treatment to patients living with blood cancers using a differentiated manufacturing process,” Susan Galbraith, MD, PhD, executive vice president of AstraZeneca’s oncology R&D."
The development of this product, and certainly more clinical trials with the product, will continue to move forward.

about the author
Audrey Burton-Bethke
Audrey is a content writer and editor for the HealthTree Foundation. She originally joined the HealthTree Foundation in 2020. Audrey loves spending time with her supportive husband, energetic four-year-old, and new baby.
More on Treatment Advances
Trending Articles




Get the Latest Multiple Myeloma Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.