First-Ever FDA Approved CAR T-Cell Therapy for R/R CLL: Liso-cel (Breyanzi)

FDA Approves CAR T-cell Therapy Liso-cel for CLL
Bristol Myers Squibb (BMS) announced on March 14th, 2024, that the FDA approved their CAR T-cell therapy product liso-cel (Breyanzi) for the treatment of CLL patients who have received at least two prior lines of treatment (including a BCL-2 inhibitor like venetoclax (Venclexta) and a BTK inhibitor like zanubrutinib [Brukinsa], acalabrutinib [Calquence], ibrutinib [Imbruvica], or pirtobrutinib [Jaypirca]). Liso-cel will be commercially available for CLL patients starting March 21st, 2024.
“CLL and SLL are currently considered incurable diseases with few treatment options in the relapsed setting that can confer complete responses, something that has historically been associated with improved long-term outcomes. The FDA approval of liso-cel in relapsed or refractory CLL and SLL after treatment with prior BTKi and BCL-2i is a remarkable breakthrough, shifting the treatment paradigm from continuous therapy with sequential regimens to overcome drug resistance, to a one-time personalized T-cell based approach that has the potential to offer patients complete and lasting remission” Dr. Tanya Siddiqi, Medical Director at the City of Hope National Medical Center (California).
In the TRANSCEND CLL 004 study, 44% of patients experienced some type of remission (partial or complete) after a single infusion of liso-cel. 20% of these cases were complete remission. On average, the patients who achieved a complete response remained in remission at the check-in point of 24 months.
How Does CAR T-cell Therapy Work?
Chimeric antigen receptor (CAR) T-cell therapy is a type of immunotherapy that helps the patient’s T cells become efficient at destroying the cancer. For some CLL patients, the one-time infusion has granted over a year of cancer-free remission.
“CAR T-cell therapies are customized for each individual patient. They are made by collecting T-cells from the patient and re-engineering them in the laboratory to produce proteins on their surface called chimeric antigen receptors, or CARs. The CARs recognize and bind to specific proteins, or antigens, on the surface of cancer cells” Cancer.gov.
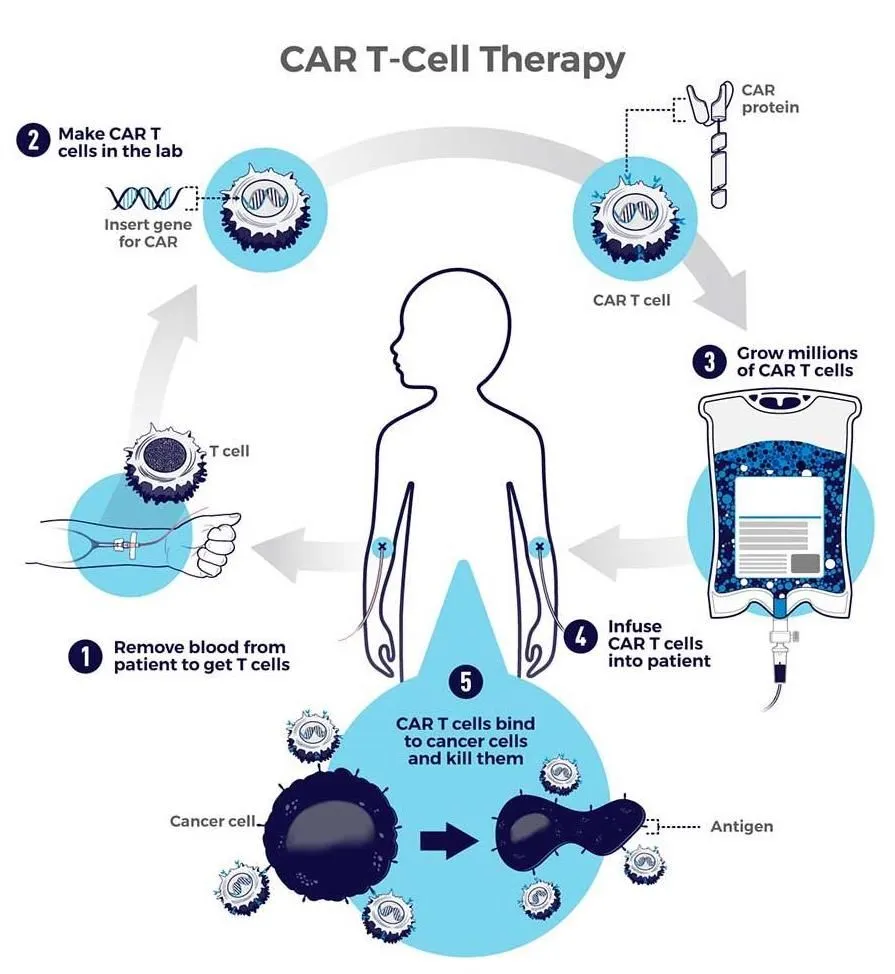
Image source: Cancer.gov
In the case of liso-cel, it will take approximately 36 days from the request until the administration of the CAR-T cell therapy.
What are the Side Effects of Liso-cel and How are They Managed?
After the infusion, you may be monitored at the infusion center for a few days to review if side-effect treatment is needed. Some patients may experience the below side effects, however, most patients quickly recover with the help of in-clinic medicines.
The possible short-term side effects of liso-cel and how they are managed:
- Cytokine release syndrome (CRS)
- Tocilizumab injection
- Corticosteroids
- Neurological (Mild: Headaches, Serious: Confusion, difficulty finding words, Severe and rare: Coma)
- Corticosteroids
- Infections
- Antibiotics, antifungals, antivirals, immunoglobulin infusions (IVIG)
- Low blood counts (cytopenias) for multiple weeks following the infusion
- Growth factor injection Nepogen to stimulate neutrophil growth
- Platelet transfusion for low platelets
- Blood transfusions for low red blood cells
- Hypersensitivity reactions
- Administer glucocorticoids, epinephrine, bronchodilators, and/or oxygen
- Fatigue
- Rest as able until resolved
- Musculoskeletal pain
- Low-dose prednisone (anti-inflammatory)
- Diarrhea
- Pepto Bismol
- Imodium
- Fiber
- Nausea
- Administer IV fluids
- Anti-nausea medication like Zofran
The long-term side-effects of liso-cel and its management:
- Decreased healthy B-cells (B-cell aplasia): The surface protein the CAR T-cells bind to on cancerous B-cells (CLL cells) to kill them is CD19. The surface protein CD19 is also on the majority of healthy B-cells. B-cells create antibodies to monitor the body’s fluids (blood and lymph fluid) that help limit invaders like bacteria or viruses from entering cells. With no healthy B-cells, patients are at an increased risk of infections.
- To compensate for B-cell aplasia, patients receive a monthly infusion of donor antibodies. The immunoglobulin infusion is often referred to as IVIG. Click here to learn more about IVIG. A patient who has received CAR T-cell therapy will need this infusion lifelong after therapy to keep their immune system healthy.
Why Would CAR T-cell Therapy Not Work?
- The CAR T-cells become exhausted, causing them to have problems multiplying in the body and attacking cancer cells. T-cell exhaustion has been proven to be caused by the build-up of too many free radicals inside the cell, exhausting the mitochondria (oxidative stress), and is reversible with antioxidants. Patients may increase the chance of CAR T-cell therapy working for them by reducing T-cell fatigue caused by oxidative stress through the consumption of antioxidant-rich foods and spices such as those listed here.
- The cancer cells may mutate, changing the membrane protein (antigen) that CAR T-cells need to bind to and making them resistant to the therapy.
In conclusion, the personalized and one-time CAR T-cell therapy infusion of liso-cel may provide relapsed/refractory CLL patients with long-term remission and manageable side effects.
Sources
- 330 Lisocabtagene Maraleucel (liso-cel) in R/R CLL/SLL: 24-Month Median Follow-up of TRANSCEND CLL 004
- Study Shows How Immune Cells Lose Their Power to Fight Tumors
- Side Effect Solutions for CLL Treatments
- U.S. FDA Approves Bristol Myers Squibb’s Breyanzi ® as the First and Only CAR T Cell Therapy for Adults with Relapsed or Refractory Chronic Lymphocytic Leukemia (CLL) or Small Lymphocytic Lymphoma (SLL)
- CAR T Cells: Engineering Patients’ Immune Cells to Treat Their Cancers
- BREYANZI U.S. Prescribing Information
- Phase 1 TRANSCEND CLL 004 study of lisocabtagene maraleucel in patients with relapsed/refractory CLL or SLL
FDA Approves CAR T-cell Therapy Liso-cel for CLL
Bristol Myers Squibb (BMS) announced on March 14th, 2024, that the FDA approved their CAR T-cell therapy product liso-cel (Breyanzi) for the treatment of CLL patients who have received at least two prior lines of treatment (including a BCL-2 inhibitor like venetoclax (Venclexta) and a BTK inhibitor like zanubrutinib [Brukinsa], acalabrutinib [Calquence], ibrutinib [Imbruvica], or pirtobrutinib [Jaypirca]). Liso-cel will be commercially available for CLL patients starting March 21st, 2024.
“CLL and SLL are currently considered incurable diseases with few treatment options in the relapsed setting that can confer complete responses, something that has historically been associated with improved long-term outcomes. The FDA approval of liso-cel in relapsed or refractory CLL and SLL after treatment with prior BTKi and BCL-2i is a remarkable breakthrough, shifting the treatment paradigm from continuous therapy with sequential regimens to overcome drug resistance, to a one-time personalized T-cell based approach that has the potential to offer patients complete and lasting remission” Dr. Tanya Siddiqi, Medical Director at the City of Hope National Medical Center (California).
In the TRANSCEND CLL 004 study, 44% of patients experienced some type of remission (partial or complete) after a single infusion of liso-cel. 20% of these cases were complete remission. On average, the patients who achieved a complete response remained in remission at the check-in point of 24 months.
How Does CAR T-cell Therapy Work?
Chimeric antigen receptor (CAR) T-cell therapy is a type of immunotherapy that helps the patient’s T cells become efficient at destroying the cancer. For some CLL patients, the one-time infusion has granted over a year of cancer-free remission.
“CAR T-cell therapies are customized for each individual patient. They are made by collecting T-cells from the patient and re-engineering them in the laboratory to produce proteins on their surface called chimeric antigen receptors, or CARs. The CARs recognize and bind to specific proteins, or antigens, on the surface of cancer cells” Cancer.gov.

Image source: Cancer.gov
In the case of liso-cel, it will take approximately 36 days from the request until the administration of the CAR-T cell therapy.
What are the Side Effects of Liso-cel and How are They Managed?
After the infusion, you may be monitored at the infusion center for a few days to review if side-effect treatment is needed. Some patients may experience the below side effects, however, most patients quickly recover with the help of in-clinic medicines.
The possible short-term side effects of liso-cel and how they are managed:
- Cytokine release syndrome (CRS)
- Tocilizumab injection
- Corticosteroids
- Neurological (Mild: Headaches, Serious: Confusion, difficulty finding words, Severe and rare: Coma)
- Corticosteroids
- Infections
- Antibiotics, antifungals, antivirals, immunoglobulin infusions (IVIG)
- Low blood counts (cytopenias) for multiple weeks following the infusion
- Growth factor injection Nepogen to stimulate neutrophil growth
- Platelet transfusion for low platelets
- Blood transfusions for low red blood cells
- Hypersensitivity reactions
- Administer glucocorticoids, epinephrine, bronchodilators, and/or oxygen
- Fatigue
- Rest as able until resolved
- Musculoskeletal pain
- Low-dose prednisone (anti-inflammatory)
- Diarrhea
- Pepto Bismol
- Imodium
- Fiber
- Nausea
- Administer IV fluids
- Anti-nausea medication like Zofran
The long-term side-effects of liso-cel and its management:
- Decreased healthy B-cells (B-cell aplasia): The surface protein the CAR T-cells bind to on cancerous B-cells (CLL cells) to kill them is CD19. The surface protein CD19 is also on the majority of healthy B-cells. B-cells create antibodies to monitor the body’s fluids (blood and lymph fluid) that help limit invaders like bacteria or viruses from entering cells. With no healthy B-cells, patients are at an increased risk of infections.
- To compensate for B-cell aplasia, patients receive a monthly infusion of donor antibodies. The immunoglobulin infusion is often referred to as IVIG. Click here to learn more about IVIG. A patient who has received CAR T-cell therapy will need this infusion lifelong after therapy to keep their immune system healthy.
Why Would CAR T-cell Therapy Not Work?
- The CAR T-cells become exhausted, causing them to have problems multiplying in the body and attacking cancer cells. T-cell exhaustion has been proven to be caused by the build-up of too many free radicals inside the cell, exhausting the mitochondria (oxidative stress), and is reversible with antioxidants. Patients may increase the chance of CAR T-cell therapy working for them by reducing T-cell fatigue caused by oxidative stress through the consumption of antioxidant-rich foods and spices such as those listed here.
- The cancer cells may mutate, changing the membrane protein (antigen) that CAR T-cells need to bind to and making them resistant to the therapy.
In conclusion, the personalized and one-time CAR T-cell therapy infusion of liso-cel may provide relapsed/refractory CLL patients with long-term remission and manageable side effects.
Sources
- 330 Lisocabtagene Maraleucel (liso-cel) in R/R CLL/SLL: 24-Month Median Follow-up of TRANSCEND CLL 004
- Study Shows How Immune Cells Lose Their Power to Fight Tumors
- Side Effect Solutions for CLL Treatments
- U.S. FDA Approves Bristol Myers Squibb’s Breyanzi ® as the First and Only CAR T Cell Therapy for Adults with Relapsed or Refractory Chronic Lymphocytic Leukemia (CLL) or Small Lymphocytic Lymphoma (SLL)
- CAR T Cells: Engineering Patients’ Immune Cells to Treat Their Cancers
- BREYANZI U.S. Prescribing Information
- Phase 1 TRANSCEND CLL 004 study of lisocabtagene maraleucel in patients with relapsed/refractory CLL or SLL

about the author
Megan Heaps
Megan joined HealthTree in 2022. She enjoys helping patients and their care partners understand the various aspects of the cancer. This understanding enables them to better advocate for themselves and improve their treatment outcomes.
More on Treatment Advances
Trending Articles
Get the Latest Chronic Lymphocytic Leukemia Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.











