The FDA Approves Columvi For Relapsed/Refractory DLBCL

Diffuse large B-cell lymphoma (DLBCL) represents one-third of all non-Hodgkin's lymphoma cases. With immediate treatment, two-thirds of patients will survive 5 years or longer. Yet, research indicates that 30-40% of DLBCL patients will relapse within the first 2 years, and 10-15% will become refractory, meaning that the patient does not respond to treatment. These statistics reveal the significant need for new and more effective DLBCL therapies.
Newly FDA-Approved Therapy
“On June 15th, 2023, the Food and Drug Administration (FDA) approved Columvi (glofitamab-gxbm) to treat adults with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) or for those with large B-cell lymphoma resulting from follicular lymphoma after two or more lines of therapy.”
How It Works
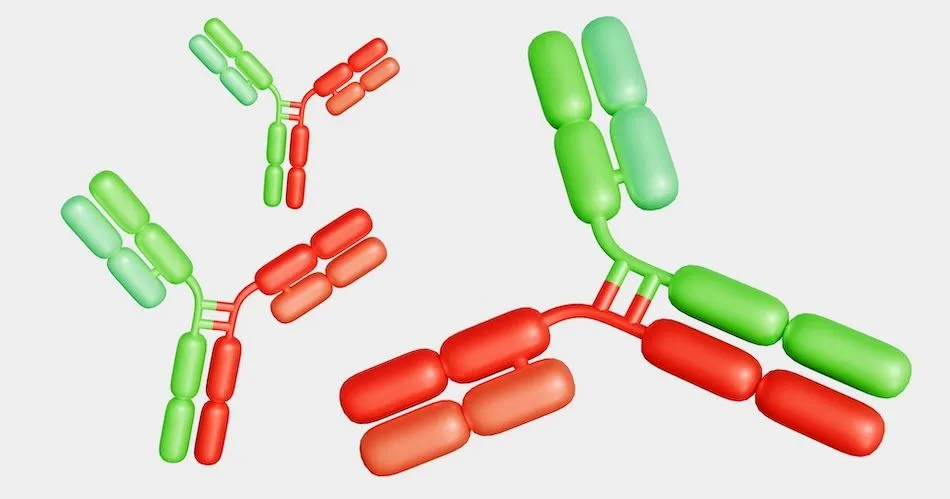
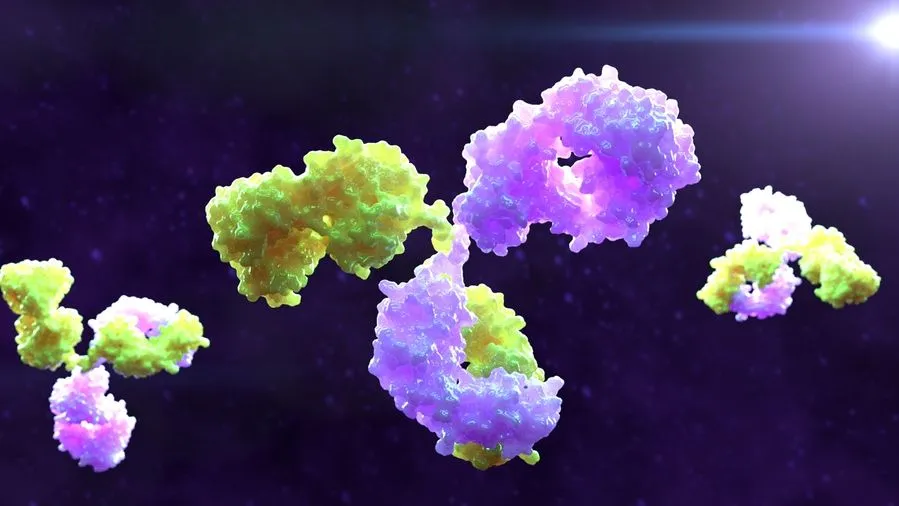
Columvi is a unique bispecific antibody that is designed to bind to 1 part of an immune cell called a T-cell and 2 parts of an immune cell called a B-cell at the same time. By binding to these cells, Columvi brings the cells close enough for the T-cell to engage with and release proteins called cytokines, which then leads to the death of cancerous B-cells. It may also harm healthy B-cells.
“Results demonstrated that patients treated with Columvi achieved durable remission, including 56% of patients who achieved an overall response (patients with a partial or complete response to treatment) and 43% obtained a complete response (the disappearance of all signs of cancer) to therapy.”
Possible side effects
- Cytokine release syndrome (CRS): can cause a fever, chills, irregular heartbeat, and difficulty breathing. Because of the serious nature of CRS, Columvi medication will be administered in a step-dose escalation protocol.
- Neurological issues: headache, confusion, trouble speaking, dizziness, tremors, fatigue
- Serious infections
- Growth in your tumor or tumor-related problems
For a detailed list of side effects, see HERE.
The most common side effects reported in the study included cytokine release syndrome (70%), fatigue (20%), musculoskeletal pain (21%) and rash (20%). Although cytokine release syndrome may be serious or life-threatening, the occurrences of this side effect in the study were generally low-grade, with 52% considered to be mild and 14% considered to be moderate.
“Experience from clinical trials demonstrates that Columvi can provide patients with relapsed or refractory diffuse large B-cell lymphoma a chance for complete remission with a fixed-duration immunotherapy and that such remissions can potentially be sustained after the end of their treatment.”
Diffuse large B-cell lymphoma (DLBCL) represents one-third of all non-Hodgkin's lymphoma cases. With immediate treatment, two-thirds of patients will survive 5 years or longer. Yet, research indicates that 30-40% of DLBCL patients will relapse within the first 2 years, and 10-15% will become refractory, meaning that the patient does not respond to treatment. These statistics reveal the significant need for new and more effective DLBCL therapies.
Newly FDA-Approved Therapy
“On June 15th, 2023, the Food and Drug Administration (FDA) approved Columvi (glofitamab-gxbm) to treat adults with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) or for those with large B-cell lymphoma resulting from follicular lymphoma after two or more lines of therapy.”
How It Works
Columvi is a unique bispecific antibody that is designed to bind to 1 part of an immune cell called a T-cell and 2 parts of an immune cell called a B-cell at the same time. By binding to these cells, Columvi brings the cells close enough for the T-cell to engage with and release proteins called cytokines, which then leads to the death of cancerous B-cells. It may also harm healthy B-cells.
“Results demonstrated that patients treated with Columvi achieved durable remission, including 56% of patients who achieved an overall response (patients with a partial or complete response to treatment) and 43% obtained a complete response (the disappearance of all signs of cancer) to therapy.”
Possible side effects
- Cytokine release syndrome (CRS): can cause a fever, chills, irregular heartbeat, and difficulty breathing. Because of the serious nature of CRS, Columvi medication will be administered in a step-dose escalation protocol.
- Neurological issues: headache, confusion, trouble speaking, dizziness, tremors, fatigue
- Serious infections
- Growth in your tumor or tumor-related problems
For a detailed list of side effects, see HERE.
The most common side effects reported in the study included cytokine release syndrome (70%), fatigue (20%), musculoskeletal pain (21%) and rash (20%). Although cytokine release syndrome may be serious or life-threatening, the occurrences of this side effect in the study were generally low-grade, with 52% considered to be mild and 14% considered to be moderate.
“Experience from clinical trials demonstrates that Columvi can provide patients with relapsed or refractory diffuse large B-cell lymphoma a chance for complete remission with a fixed-duration immunotherapy and that such remissions can potentially be sustained after the end of their treatment.”

about the author
Lisa Foster
Lisa Foster is a mom of 3 daughters and 1 perfect grandchild, a puzzle lover, writer and HealthTree advocate. She believes in the mission of the foundation and the team that builds it forward. She calls Houston, Texas home.
More on Clinical Trials
Trending Articles
Get the Latest Large B Cell Lymphoma Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.