CLL Patients - What is CAR T-Cell Therapy?
Chimeric antigen receptor (CAR) T-cell therapy is a type of immunotherapy that helps the patient’s T-cells become efficient at destroying the cancer. For some CLL patients, it has cured their cancer.
Although CAR T-cell therapy is FDA-approved for a few other types of blood cancers, CAR T-cell therapy is not yet FDA-approved for the treatment of CLL. To help get it approved for CLL treatment, there are many clinical trials underway that CLL patients have joined and are continuing to join. For information about finding and joining CAR T-cell therapy clinical trials, see here: Clinical trial finder for CLL patients (in the "Search by Keyword" bar near the upper left-hand side of the page, type CAR T. You can then filter based on location and recruiting clinical trials).
How it Works
“CAR T-cell therapies are customized for each individual patient. They are made by collecting T-cells from the patient and re-engineering them in the laboratory to produce proteins on their surface called chimeric antigen receptors, or CARs. The CARs recognize and bind to specific proteins, or antigens, on the surface of cancer cells” (cancer.gov).
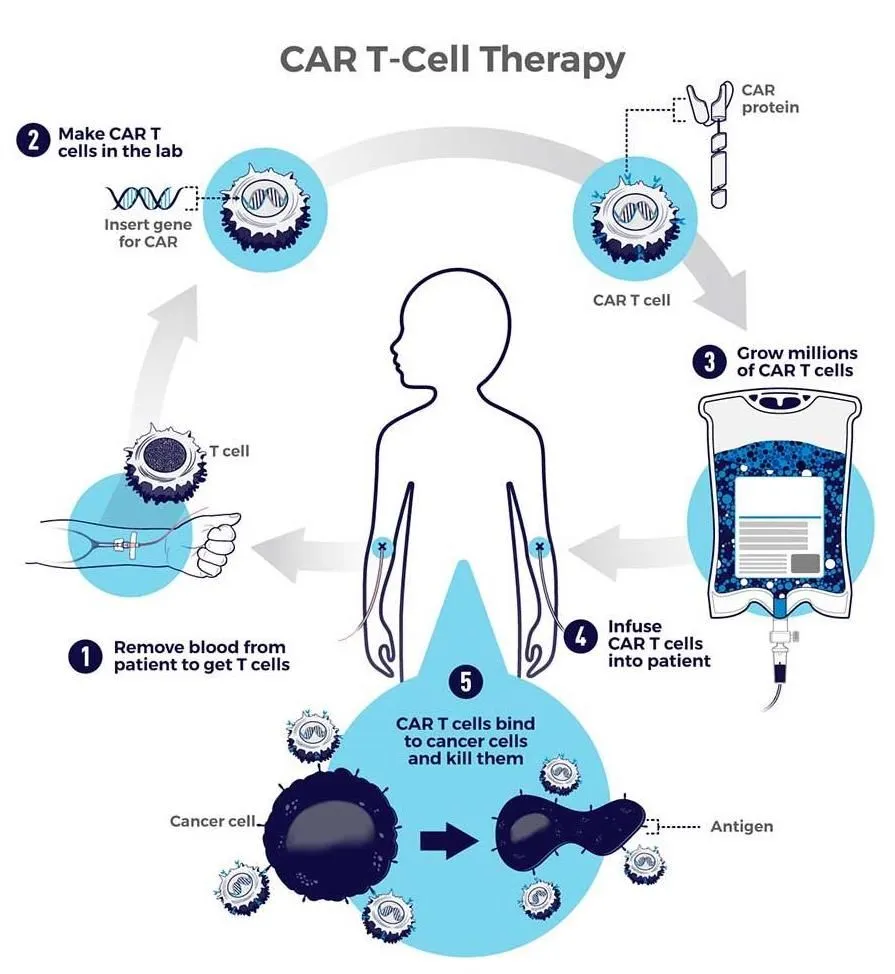
Image source: cancer.gov
Patients are monitored for a few days at the clinic following treatment to review if side-effect treatment is needed related to cytokine release syndrome or neurological symptoms. Some patients may experience these side effects however most patients quickly recover with the help of in-clinic side-effect solution medicines. Researchers are also continuing to study more ways to help CAR T-cell therapy be more effective with fewer side effects.
Why would CAR T-cell therapy not work?
- CAR T-cells may have problems multiplying inside the body
- The CAR T-cells may become exhausted (researchers believe this may be linked to the proteins that turn genes on and off. A study showed that T-cell exhaustion was linked to excess oxidative stress and adding more antioxidants reduced the T-cells' exhaustion allowing them to work better and last longer to destroy cancer cells. See here to learn more about antioxidants and T-cells: Strengthen Cancer-killing T-cells with Antioxidants)
- The cancer cells may mutate changing the membrane protein (antigen) that CAR T-cells need to bind to
Talk with your CLL specialist to see if joining a CAR T-cell clinical trial is right for you. Need help finding a CLL specialist? Visit HealthTree’s CLL Specialist Directory.
Chimeric antigen receptor (CAR) T-cell therapy is a type of immunotherapy that helps the patient’s T-cells become efficient at destroying the cancer. For some CLL patients, it has cured their cancer.
Although CAR T-cell therapy is FDA-approved for a few other types of blood cancers, CAR T-cell therapy is not yet FDA-approved for the treatment of CLL. To help get it approved for CLL treatment, there are many clinical trials underway that CLL patients have joined and are continuing to join. For information about finding and joining CAR T-cell therapy clinical trials, see here: Clinical trial finder for CLL patients (in the "Search by Keyword" bar near the upper left-hand side of the page, type CAR T. You can then filter based on location and recruiting clinical trials).
How it Works
“CAR T-cell therapies are customized for each individual patient. They are made by collecting T-cells from the patient and re-engineering them in the laboratory to produce proteins on their surface called chimeric antigen receptors, or CARs. The CARs recognize and bind to specific proteins, or antigens, on the surface of cancer cells” (cancer.gov).

Image source: cancer.gov
Patients are monitored for a few days at the clinic following treatment to review if side-effect treatment is needed related to cytokine release syndrome or neurological symptoms. Some patients may experience these side effects however most patients quickly recover with the help of in-clinic side-effect solution medicines. Researchers are also continuing to study more ways to help CAR T-cell therapy be more effective with fewer side effects.
Why would CAR T-cell therapy not work?
- CAR T-cells may have problems multiplying inside the body
- The CAR T-cells may become exhausted (researchers believe this may be linked to the proteins that turn genes on and off. A study showed that T-cell exhaustion was linked to excess oxidative stress and adding more antioxidants reduced the T-cells' exhaustion allowing them to work better and last longer to destroy cancer cells. See here to learn more about antioxidants and T-cells: Strengthen Cancer-killing T-cells with Antioxidants)
- The cancer cells may mutate changing the membrane protein (antigen) that CAR T-cells need to bind to
Talk with your CLL specialist to see if joining a CAR T-cell clinical trial is right for you. Need help finding a CLL specialist? Visit HealthTree’s CLL Specialist Directory.

about the author
Megan Heaps
Megan joined HealthTree in 2022. She enjoys helping patients and their care partners understand the various aspects of the cancer. This understanding enables them to better advocate for themselves and improve their treatment outcomes.
More on Treatment Advances
Trending Articles
Get the Latest Chronic Lymphocytic Leukemia Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.