ASH 2023: Predicting Drug Toxicity in CAR T-Cell Therapy Recipients

In the world of cellular therapy, Dr. Roni Shouval, an attending physician and scientist at Memorial Sloan Kettering Cancer Center, is exploring efficacy (producing the desired result) and toxicity. At the forefront of groundbreaking research, Dr. Shouval, on behalf of the Center for International Blood and Marrow Transplant Research (CIBMTR), presented a pivotal study at the 2023 American Society of Hematology (ASH) conference. The focus is on toxicities associated with chimeric antigen receptor (CAR) T-cell therapy, a revolutionary approach transforming the landscape of care for blood cancers.
CAR T-Cell Therapy
CAR T-cell therapy is an immunotherapy often used in blood cancers such as leukemia, lymphoma, and multiple myeloma. The following table shows current FDA approved CAR T-cell treatments:

This treatment uses the power of the patient's own immune system. This is a personalized approach collecting T cells from the patient, genetically modifying those cells in a lab, then returning them back to the body. CAR T-cell therapy creates a strong immune response, leading to the destruction of cancer cells. Specifically, CD19 CAR T-cell therapy targets the CD19 protein on B-cells in patients with B-cell lymphomas like diffuse large B-cell lymphoma, follicular lymphoma, mantle cell lymphoma, chronic lymphocytic leukemia, and acute lymphocytic leukemia.
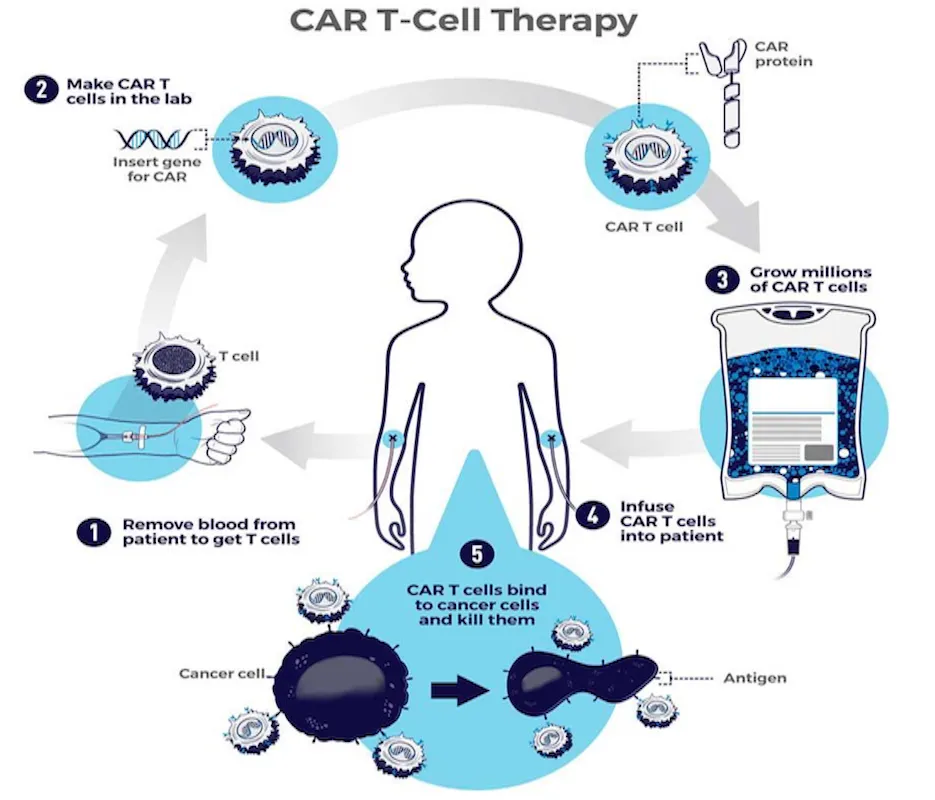
CAR T-cell therapy represents a paradigm shift in treating patients, especially those with lymphomas not responding (refractory) to traditional chemotherapy. While it opens doors to longer remissions (full or partial reduction in signs and symptoms) and potential cures, Dr. Shouval mentions the downside—significant toxicities are linked to this therapy. The two main toxicities are cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS).
CRS and ICANS
CRS, a systemic response triggered by the infusion of CAR T-cells, presents as a widespread inflammatory reaction. Patients may experience fever, a drop in blood pressure, and respiratory difficulties, with severe cases posing life-threatening complications. On the other hand, ICANS presents a spectrum of neurotoxic manifestations, ranging from mild tremors to coma or seizures, sometimes leading to fatal outcomes. Dr. Shouval mentions ICANS as the broader term for neurotoxicity.
Side Effects of Axicabtagene Ciloleucel and Tisagenlecleucel
In the comprehensive analysis presented at ASH, Dr. Shouval discussed one of the largest cohorts to date, consisting of 1,916 patients from 97 medical centers across the United States. Focusing on CD19-directed CAR T-cell therapy for large B-cell lymphoma, the study aimed to understand the incidence (occurrence) and predictors of CRS and ICANS in this patient population.
Regarding toxicity occurrence, the following was determined:
- A significant proportion of patients—75%—developed low-grade CRS (grade one or two), indicating manageable side effects
- Severe CRS, categorized as grade three or higher, affected only 9% of the group, offering reassurance regarding the therapy's safety profile
- 56% of patients experienced ICANS
- 20% faced severe ICANS
- Almost all patients who developed ICANS also experienced CRS (98%)
Regarding toxicity predictors, the following was determined:
- The type of CAR T-cell product used played a significant role, with axicabtagene ciloleucel (axi-cel) being more likely to induce CRS and ICANS compared to tisagenlecleucel (tisa-cel)
- Risk factors included older age, lower performance status at the time of cellular therapy administration, high LDH at time of cellular therapy administration, and gender, with women being at higher risk
Regarding the impact of toxicities on patient survival, the following was determined:
- Overall CRS, early onset CRS, and overall ICANS were not linked to higher mortality (death rates)
- Severe grades (>3) of CRS and ICANS, along with early onset ICANS (less than 7 days from start of treatment), were associated with increased mortality (death rates)
- Severe CRS significantly impacted survival, as shown in the analysis at 30 days from infusion
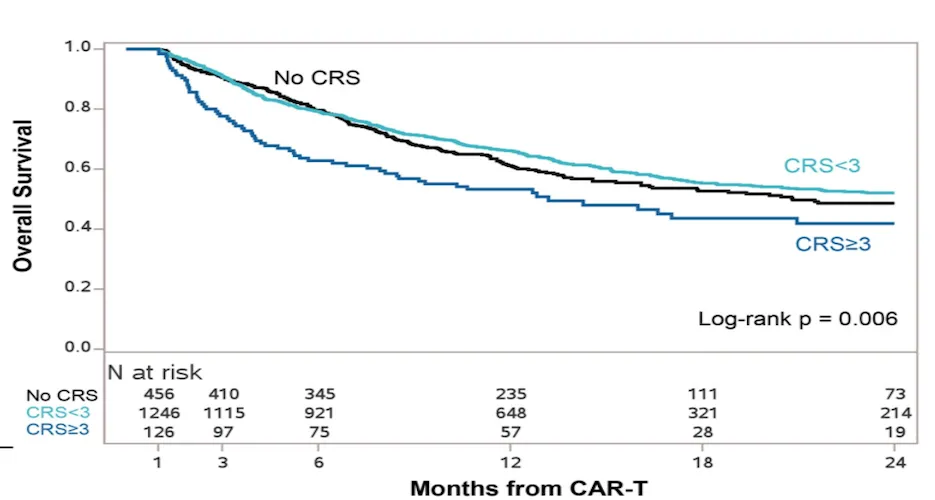
Patients experiencing low-grade or no CRS exhibited longer survival rates compared to those suffering with severe manifestations of these complications.
Prospective CAR-T Cell Therapy Safety
In conclusion, Dr. Roni Shouval's analysis suggests while CAR T-cell therapy offers new hope to those with blood cancer, CRS and ICANS complications require a vigilant approach. Providers must be aware of risk factors and consistently monitor patients for toxicity. As the scientific community continues to unravel the complexities of cellular therapies, Dr. Shouval emphasizes the need for preventative measures to curb these severe toxicities and mitigate complications.
In the world of cellular therapy, Dr. Roni Shouval, an attending physician and scientist at Memorial Sloan Kettering Cancer Center, is exploring efficacy (producing the desired result) and toxicity. At the forefront of groundbreaking research, Dr. Shouval, on behalf of the Center for International Blood and Marrow Transplant Research (CIBMTR), presented a pivotal study at the 2023 American Society of Hematology (ASH) conference. The focus is on toxicities associated with chimeric antigen receptor (CAR) T-cell therapy, a revolutionary approach transforming the landscape of care for blood cancers.
CAR T-Cell Therapy
CAR T-cell therapy is an immunotherapy often used in blood cancers such as leukemia, lymphoma, and multiple myeloma. The following table shows current FDA approved CAR T-cell treatments:

This treatment uses the power of the patient's own immune system. This is a personalized approach collecting T cells from the patient, genetically modifying those cells in a lab, then returning them back to the body. CAR T-cell therapy creates a strong immune response, leading to the destruction of cancer cells. Specifically, CD19 CAR T-cell therapy targets the CD19 protein on B-cells in patients with B-cell lymphomas like diffuse large B-cell lymphoma, follicular lymphoma, mantle cell lymphoma, chronic lymphocytic leukemia, and acute lymphocytic leukemia.

CAR T-cell therapy represents a paradigm shift in treating patients, especially those with lymphomas not responding (refractory) to traditional chemotherapy. While it opens doors to longer remissions (full or partial reduction in signs and symptoms) and potential cures, Dr. Shouval mentions the downside—significant toxicities are linked to this therapy. The two main toxicities are cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS).
CRS and ICANS
CRS, a systemic response triggered by the infusion of CAR T-cells, presents as a widespread inflammatory reaction. Patients may experience fever, a drop in blood pressure, and respiratory difficulties, with severe cases posing life-threatening complications. On the other hand, ICANS presents a spectrum of neurotoxic manifestations, ranging from mild tremors to coma or seizures, sometimes leading to fatal outcomes. Dr. Shouval mentions ICANS as the broader term for neurotoxicity.
Side Effects of Axicabtagene Ciloleucel and Tisagenlecleucel
In the comprehensive analysis presented at ASH, Dr. Shouval discussed one of the largest cohorts to date, consisting of 1,916 patients from 97 medical centers across the United States. Focusing on CD19-directed CAR T-cell therapy for large B-cell lymphoma, the study aimed to understand the incidence (occurrence) and predictors of CRS and ICANS in this patient population.
Regarding toxicity occurrence, the following was determined:
- A significant proportion of patients—75%—developed low-grade CRS (grade one or two), indicating manageable side effects
- Severe CRS, categorized as grade three or higher, affected only 9% of the group, offering reassurance regarding the therapy's safety profile
- 56% of patients experienced ICANS
- 20% faced severe ICANS
- Almost all patients who developed ICANS also experienced CRS (98%)
Regarding toxicity predictors, the following was determined:
- The type of CAR T-cell product used played a significant role, with axicabtagene ciloleucel (axi-cel) being more likely to induce CRS and ICANS compared to tisagenlecleucel (tisa-cel)
- Risk factors included older age, lower performance status at the time of cellular therapy administration, high LDH at time of cellular therapy administration, and gender, with women being at higher risk
Regarding the impact of toxicities on patient survival, the following was determined:
- Overall CRS, early onset CRS, and overall ICANS were not linked to higher mortality (death rates)
- Severe grades (>3) of CRS and ICANS, along with early onset ICANS (less than 7 days from start of treatment), were associated with increased mortality (death rates)
- Severe CRS significantly impacted survival, as shown in the analysis at 30 days from infusion

Patients experiencing low-grade or no CRS exhibited longer survival rates compared to those suffering with severe manifestations of these complications.
Prospective CAR-T Cell Therapy Safety
In conclusion, Dr. Roni Shouval's analysis suggests while CAR T-cell therapy offers new hope to those with blood cancer, CRS and ICANS complications require a vigilant approach. Providers must be aware of risk factors and consistently monitor patients for toxicity. As the scientific community continues to unravel the complexities of cellular therapies, Dr. Shouval emphasizes the need for preventative measures to curb these severe toxicities and mitigate complications.

about the author
Kat Richardson
Kat is from Lehi, Utah and is the education manager for lymphoma. She has worked in healthcare for a decade now, and earned her degree in community health education and promotion. Kat is passionate about disease prevention as well as improving quality of life and health equity. She enjoys reading, hiking, baking, ice skating, gardening, time with her family and friends, and most of all, spoiling her nieces and nephew.
More on Conferences
Trending Articles
Get the Latest Large B Cell Lymphoma Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.