Study of Evorpacept and Azacitidine for High-Risk MDS

Myelodysplastic syndromes (MDS) are a group of diverse bone marrow disorders in which the bone marrow does not produce enough healthy blood cells. It is considered a type of blood cancer.
MDS cells express CD47 to avoid detection by healthy immune cells, which are in charge of eliminating cancer cells.
ALX Oncology is developing a next-generation checkpoint inhibitor called evoparcept. It is designed to bind and block CD47 and avoid the side effects seen with other CD47-blocking therapies. When it binds to CD47, evoparcept helps signal the immune response against lymphoma cells.
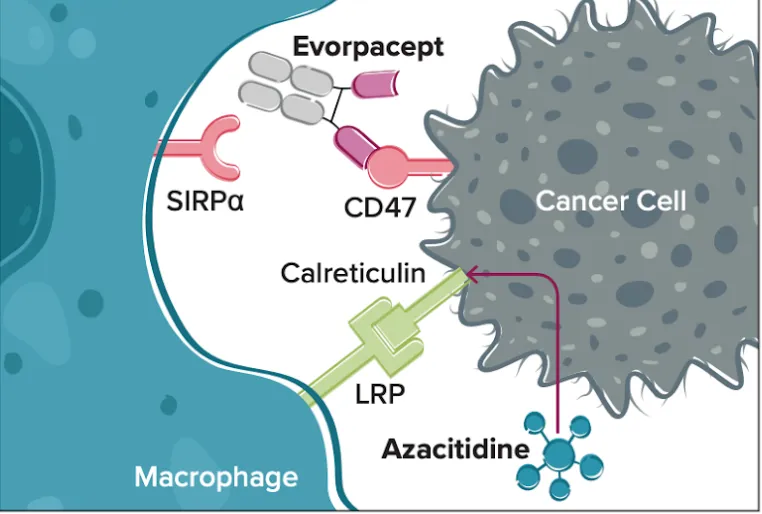
Image retrieved from: ALX Oncology (2024) A Study of Evorpacept (ALX148) With Azacitidine for Higher Risk Myelodysplastic Syndrome (ASPEN-02). The image illustrates how CD47 protein expressed by cancer cells is blocked by evorpacept, that way, a receptor in macrophages (immune system cells that eliminate cancer cells or infected cells), can identify the cancer cell that was camouflaging as a “good” cell with CD47, and in combination with azacitidine, the bind becomes stronger since it aids another receptor acting in this immune response.
Evorpacept helps signal macrophages, an immune cell that “eats” sick or cancerous cells. By covering the CD47 expressed by cancer cells, it removes their “camouflage” ability, which makes macrophages avoid them. That way, it’s easier to identify cancer cells that need to be eliminated.
The ASPEN-02 is a phase 1/2 clinical study that evaluates evorpacept in combination with azacitidine for the treatment of patients with high-risk MDS. It is not currently enrolling patients.
What Do We Know About Evorpacept in MDS So Far?
Investigators shared initial data of evorpacept combined with azacitidine to treat 65 patients with MDS: 52 with newly diagnosed high-risk MDS and 13 patients with relapsed disease.
Best results have been reported in the high-risk, newly diagnosed group:
- 40% of patients achieved a response, in some cases with a significant reduction of blast cells in the bone marrow.
- Half of the patients who responded were still in remission after 8 months.
- The median overall survival was 16.6 months.
Evorpacept can enhance the anti-cancer activity of azacitidine with minimal additional toxicity:
- Adverse effects were reported in all patients (65); however, they were generally asymptomatic or mild.
- None of the patients discontinued treatment due to side effects
- The most frequent side effects experienced were diarrhea, nausea, vomiting, a decrease in white blood cell counts (neutrophils), and reactions to the therapy vein infusion.
- Importantly, of the 26 patients who were transfusion-dependent at the beginning of treatment, 10 patients achieved transfusion independence that lasted from 8 to 48 weeks.
New Treatment Options are Urgently Needed for High-Risk MDS Patients
High-risk MDS patients have limited treatment options. Treatments manage symptoms but often don't stop the disease from progressing to leukemia. Developing new therapies is crucial, not just for symptom relief, but to change the course of the disease entirely.
Clinical research plays a vital role in developing these groundbreaking therapies. By participating in clinical trials, high-risk MDS patients can contribute to advancements that could lead to better treatments, improved survival rates, and a higher quality of life for themselves and future patients.
With our clinical trial finder found within HealthTree Cure Hub, you can find up-to-date information on clinical trials looking for participants in your area!
If you want to keep reading about MDS treatment options, make sure you check the previous articles published on the HealthTree for MDS site:
- The Role Of Hypomethylating Agents
- ASH 2023: Venetoclax Combined With Azacitidine For High-Risk MDS
- A New Treatment Option Being Investigated For High-Risk MDS And AML Patients
Sources:
- ALX Oncology Announces Initial Data from ASPEN-02, the Ongoing Phase 1 / 2 Study of Evorpacept in Combination with Azacitidine, Demonstrating Safety and Preliminary Activity in Patients with Myelodysplastic Syndrome
- ASPEN-02: A Phase 1 Study of Azacitidine in Combination with Evorpacept for Higher-Risk Myelodysplastic Syndrome. 2024 American Association for Cancer Research (“AACR”) Annual Meeting, Abstract #CT060.
Myelodysplastic syndromes (MDS) are a group of diverse bone marrow disorders in which the bone marrow does not produce enough healthy blood cells. It is considered a type of blood cancer.
MDS cells express CD47 to avoid detection by healthy immune cells, which are in charge of eliminating cancer cells.
ALX Oncology is developing a next-generation checkpoint inhibitor called evoparcept. It is designed to bind and block CD47 and avoid the side effects seen with other CD47-blocking therapies. When it binds to CD47, evoparcept helps signal the immune response against lymphoma cells.
Image retrieved from: ALX Oncology (2024) A Study of Evorpacept (ALX148) With Azacitidine for Higher Risk Myelodysplastic Syndrome (ASPEN-02). The image illustrates how CD47 protein expressed by cancer cells is blocked by evorpacept, that way, a receptor in macrophages (immune system cells that eliminate cancer cells or infected cells), can identify the cancer cell that was camouflaging as a “good” cell with CD47, and in combination with azacitidine, the bind becomes stronger since it aids another receptor acting in this immune response.
Evorpacept helps signal macrophages, an immune cell that “eats” sick or cancerous cells. By covering the CD47 expressed by cancer cells, it removes their “camouflage” ability, which makes macrophages avoid them. That way, it’s easier to identify cancer cells that need to be eliminated.
The ASPEN-02 is a phase 1/2 clinical study that evaluates evorpacept in combination with azacitidine for the treatment of patients with high-risk MDS. It is not currently enrolling patients.
What Do We Know About Evorpacept in MDS So Far?
Investigators shared initial data of evorpacept combined with azacitidine to treat 65 patients with MDS: 52 with newly diagnosed high-risk MDS and 13 patients with relapsed disease.
Best results have been reported in the high-risk, newly diagnosed group:
- 40% of patients achieved a response, in some cases with a significant reduction of blast cells in the bone marrow.
- Half of the patients who responded were still in remission after 8 months.
- The median overall survival was 16.6 months.
Evorpacept can enhance the anti-cancer activity of azacitidine with minimal additional toxicity:
- Adverse effects were reported in all patients (65); however, they were generally asymptomatic or mild.
- None of the patients discontinued treatment due to side effects
- The most frequent side effects experienced were diarrhea, nausea, vomiting, a decrease in white blood cell counts (neutrophils), and reactions to the therapy vein infusion.
- Importantly, of the 26 patients who were transfusion-dependent at the beginning of treatment, 10 patients achieved transfusion independence that lasted from 8 to 48 weeks.
New Treatment Options are Urgently Needed for High-Risk MDS Patients
High-risk MDS patients have limited treatment options. Treatments manage symptoms but often don't stop the disease from progressing to leukemia. Developing new therapies is crucial, not just for symptom relief, but to change the course of the disease entirely.
Clinical research plays a vital role in developing these groundbreaking therapies. By participating in clinical trials, high-risk MDS patients can contribute to advancements that could lead to better treatments, improved survival rates, and a higher quality of life for themselves and future patients.
With our clinical trial finder found within HealthTree Cure Hub, you can find up-to-date information on clinical trials looking for participants in your area!
If you want to keep reading about MDS treatment options, make sure you check the previous articles published on the HealthTree for MDS site:
- The Role Of Hypomethylating Agents
- ASH 2023: Venetoclax Combined With Azacitidine For High-Risk MDS
- A New Treatment Option Being Investigated For High-Risk MDS And AML Patients
Sources:
- ALX Oncology Announces Initial Data from ASPEN-02, the Ongoing Phase 1 / 2 Study of Evorpacept in Combination with Azacitidine, Demonstrating Safety and Preliminary Activity in Patients with Myelodysplastic Syndrome
- ASPEN-02: A Phase 1 Study of Azacitidine in Combination with Evorpacept for Higher-Risk Myelodysplastic Syndrome. 2024 American Association for Cancer Research (“AACR”) Annual Meeting, Abstract #CT060.

about the author
Jimena Vicencio
Jimena is an International Medical Graduate and a member of the HealthTree Writing team. She has a passion for languages and is currently learning Japanese. In her free time, she loves playing with her cats. Jimena is also pursuing a bachelor's degree in journalism.
More on Clinical Trials
Get the Latest Myelodysplastic Syndromes Updates, Delivered to You.
By subscribing to the HealthTree newsletter, you'll receive the latest research, treatment updates, and expert insights to help you navigate your health.
Together we care.
Together we cure.